Computer Systems Validation, Part 1: Planning
Course Summary
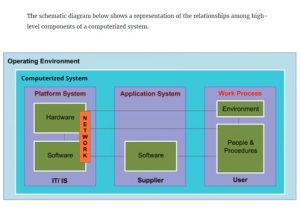
In the medicines and healthcare products industries, computerised systems used in automated manufacturing or laboratory processes to which Good Manufacturing Practice requirements apply need to be validated. This module describes the planning of such validation. It follows the work of a pharmaceutical company’s team as they validate the dispensary control system for a new production line.
£99.00 Original price was: £99.00.£79.00Current price is: £79.00. exc. VAT
Purchasing Information
When you have completed your course order, Zenosis will finalise the setup of your course materials and contact you on the email address that you provide in your order. You can expect this process to be completed within one business day (using the UK business calendar) of completing your payment.
It is therefore essential that you use your real email address for your order, or indicate in the purchase notes the email address to be used for the course set-up, and check that any messages from Zenosis or grapl are not lost in your junk or spam folder.
You will have access to the course module(s) for a period of 180 days after your purchase is complete.
Detailed Course Information
• Define computer systems validation
• Outline criteria for selecting systems to be validated and for initial estimation of the degree of validation required
• Access important guidance documents by industry bodies and regulatory authorities
• Identify the phases of the computer systems lifecycle and describe the activities that are performed in each phase
• Describe considerations influencing validation strategy
• Assess software suppliers and their products
• Outline the contents of a validation plan
Manufacturing personnel in the pharma/biotech, dietary supplement, and medical devices industries need to understand the principles and practice of computerised system validation, as set out in this module. In particular, the module provides essential learning for engineering, information and communication technology, production, and quality management personnel in the pharmaceutical industry.
• Introduction
– This session defines computer system validation and specifies its benefits. It identifies, in general terms, which systems need to be validated. It identifies sources of guidance from industry bodies and regulatory authorities, and it discusses the importance of protection of data integrity.
• The planning phase
– This session identifies the phases of the computer systems lifecycle, and outlines the activities that are performed in the planning phase. It specifies the purposes of a User Requirements Specification and a traceability matrix.
• Validation strategy and plan
– This session specifies criteria for regulatory assessment. It outlines FDA requirements on electronic records and electronic signatures. It describes in detail how to assess software suppliers and their products. It sets out principles of risk management. Finally, it outlines the contents of a validation plan, including change management.
• Assessment
– The assessment tests the learner’s assimilation of the module’s content.
